These days I was surfing on the internet and found this interesting title and I immediately wanted to share with you :) This was prepared thinking on the Halloween thematic.
You probably already have a pretty good idea of the reasons behind the red colouration of human blood that fake blood mimics. However, red is not the only blood colour available – it also comes in blue, green, violet, and even colourless varieties – and this is a result of the specific chemicals that make up blood in different organisms.
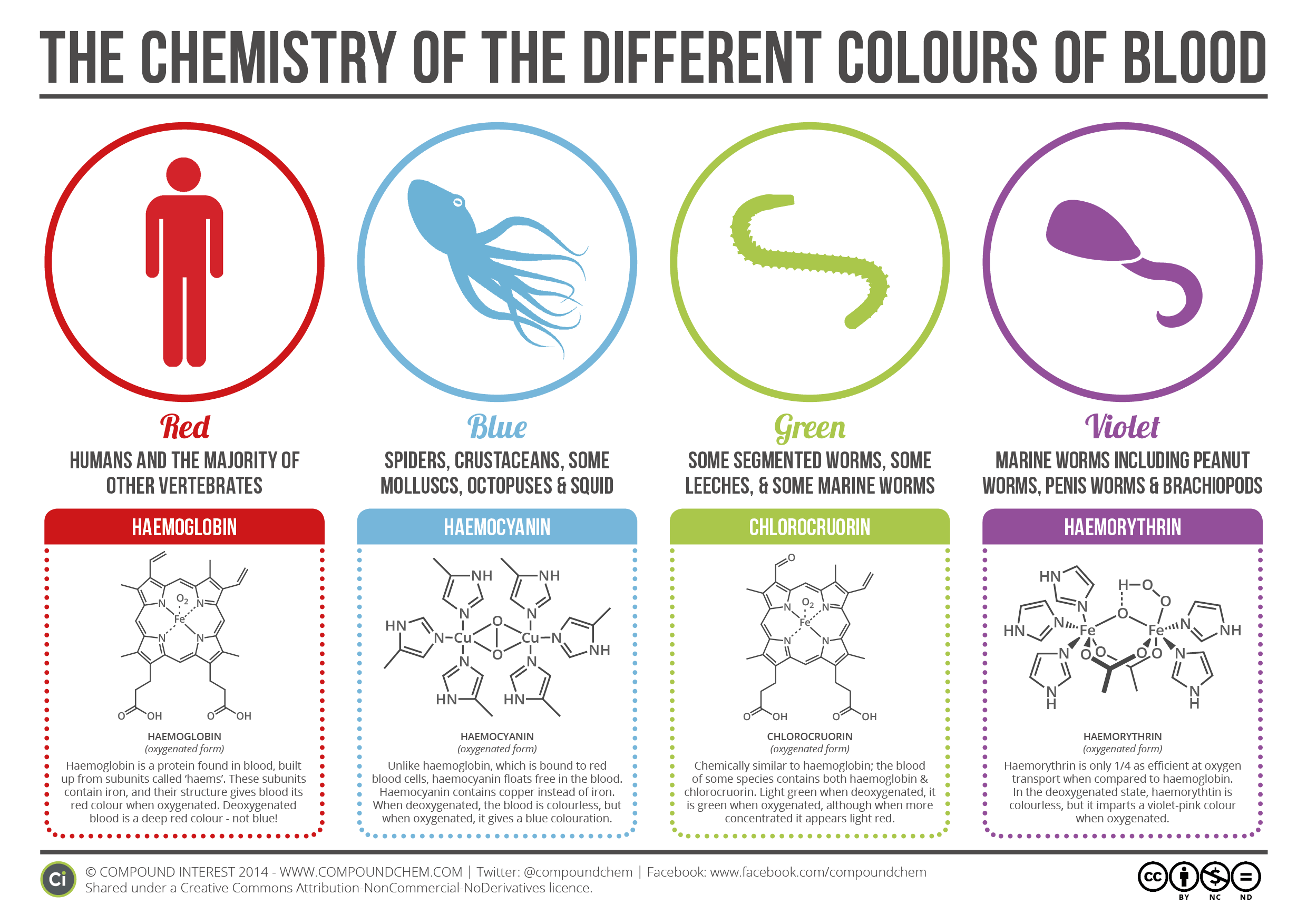
So, let’s start with what we already know. Most people will have learnt that human blood, as well as that of most other vertebrates, is red as a result of haemoglobin, a large protein found in red blood cells which contains iron atoms within its structure. Haemoglobin is what’s known as a respiratory pigment, and it plays a vital role in the body, ferrying oxygen around the body to your cells and helping carbon dioxide back to the lungs where it can be exhaled. The large protein consists of four smaller units known individually as haems, each of which contains an iron atom. This can ‘bind’ to oxygen, giving red blood cells their oxygen transporting ability.
The iron atoms are also responsible for haemoglobin’s colour. The individual haems are conjugated molecules – they have lots of alternating double and single bonds between carbon atoms in their structure – and this conjugation causes them to absorb light wavelengths in the visible portion of the spectrum, leading to a coloured appearance. The presence of the iron atom modifies this absorption slightly, and as such haemoglobin is a red colour when oxygenated, and a slightly darker red when deoxygenated.
It’s a commonly believed myth that deoxygenated blood is blue – after all, if you look through your skin at any of your veins, which carry deoxygenated blood away from your body’s cells, they have a definite blue-grey hue. However, this appearance is in fact caused by the interaction of light with both the blood and the skin and tissue covering the veins. There’s a detailed look at the slightly more complex reasons why veins appear blue, despite being red, in this paper examining the question.
There are some creatures, however, for whom blue blood is the norm. Crustaceans, spiders, squid, octopuses, and some molluscs all have blue blood as a result of having a different respiratory pigment. Rather than haemoglobin, these creatures use a protein called haemocyanin to transport oxygen. The differing structure of the pigment, as well as the incorporation of copper atoms instead of iron, leads to the blood being colourless when deoxygenated, and blue when oxygenated. They also bind to oxygen in a different manner to haemoglobin, with two copper atoms binding to each oxygen molecule.
It doesn’t stop there; green blood, too, is possible, in some species of worms and leeches. This is an interesting one, in that the individual units of chlorocruorin, the protein leading to a green blood colouration, are actually very similar in appearance to haemoglobin. In fact, they’re near identical – the only different is an aldehyde group in the place of a vinyl group in the chemical structure (although the name might suggest otherwise, chlorocruorin doesn’t contain any chlorine atoms).
Despite this minor difference, a noticeable colour change is the result – deoxygenated blood containing chlorocruorin is a light green colour, and a slightly darker green when oxygenated. Oddly, in concentrated solutions, it takes on a light red colour. A number of organisms that have chlorocruorin in their blood also have haemoglobin present as well, resulting in an overall red colouration.
Chlorocruorin isn’t always necessary for green blood, however, as the green-blooded skink lizard illustrates. This lizard is found in New Guinea, and despite its blood containing haemoglobin like other vertebrates, its blood is a distinctive green colour. The colour is due to a difference in how they recycle haemoglobin. Humans recycle haemoglobin in the liver, by breaking it down first into biliverdin, and then bilirubin. The lizards, however, aren’t capable of breaking down biliverdin any further, so it accumulates in their blood, giving a green colour intense enough to overpower the red colour of haemoglobin.
Finally, violet blood is also possible, albeit in a limited range of marine worms (including the rather unfortunately named penis worms). This colour is caused by yet another different respiratory pigment, this time one called haemorythrin. Haemorythrin contains individual units which themselves contain iron atoms; when deoxygenated, the blood is colourless, but when oxygenated it is a bright violet-pink. Like most of the other respiratory pigments, it’s a lot less efficient than haemoglobin, in some cases only having around a quarter of the oxygen carrying capacity.
What’s perhaps most interesting about the varying colours of blood is that it showcases evolution coming up with different solutions to the same problem – in this case, transporting oxygen. It’s funny to think that, if our blood incorporated copper-containing respiration pigments instead of iron, we might all be slapping on a different colour fake blood instead of red!
That's interesting, but I really don't understand those chemical things xD
ReplyDeleteThanks for sharing :)